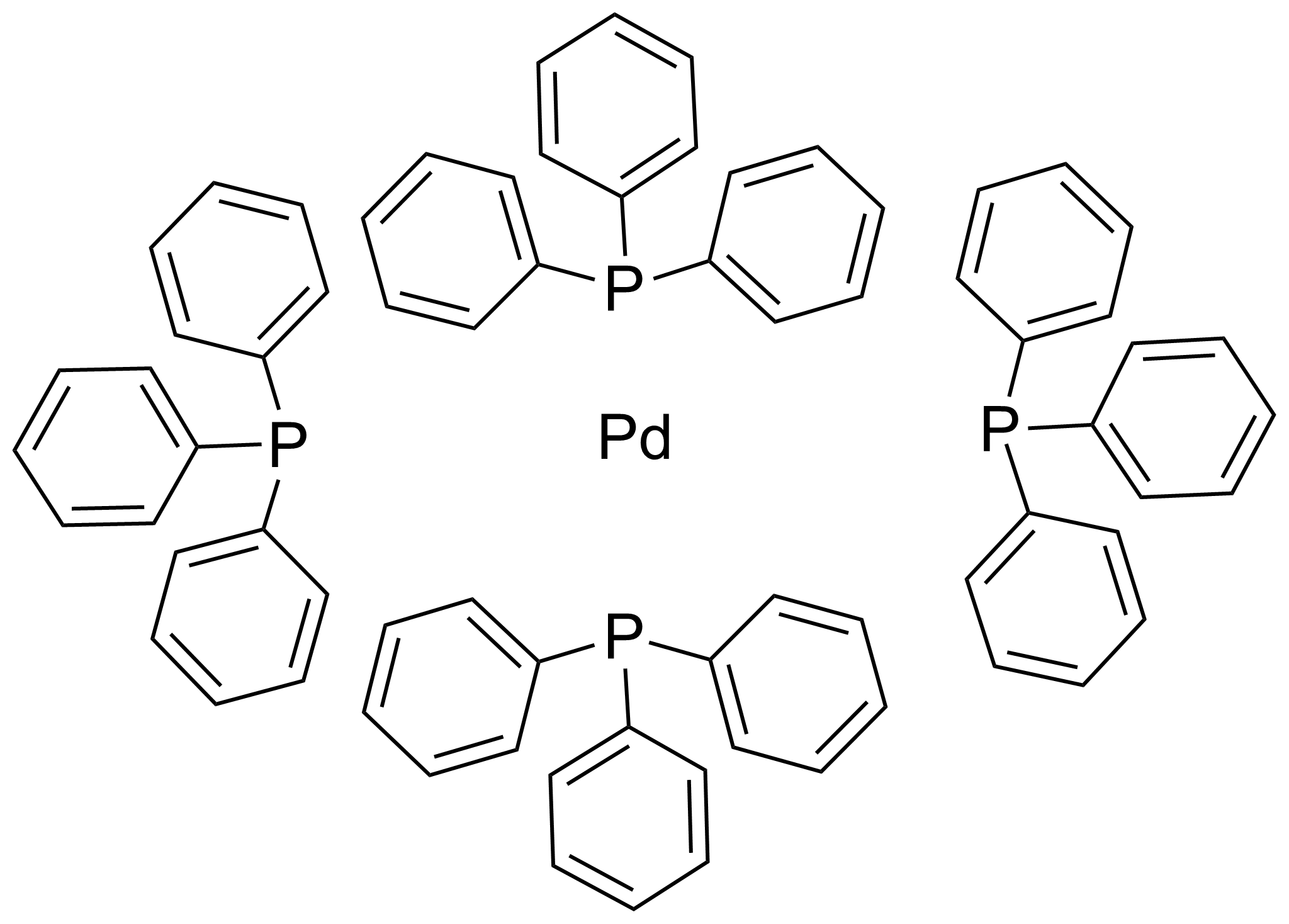
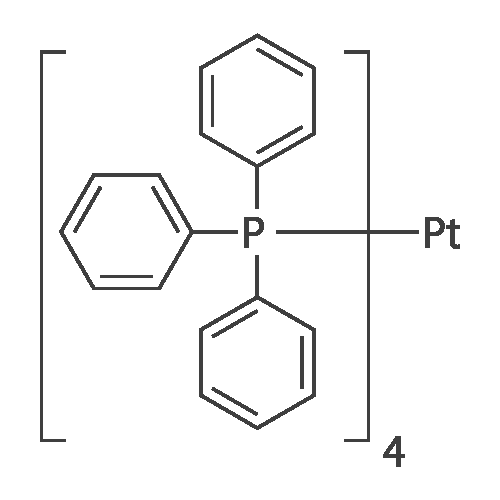
![14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/14221-01-3.png)
14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

Synthesis of pinacol arylboronates via cross-coupling reaction of bis(pinacolato)diboron with chloroarenes catalyzed by palladium(0)–tricyclohexylphosphine complexes - ScienceDirect

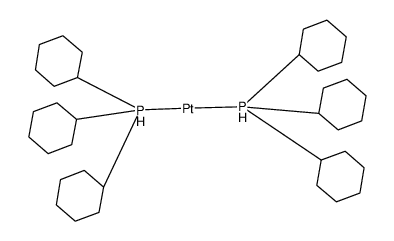
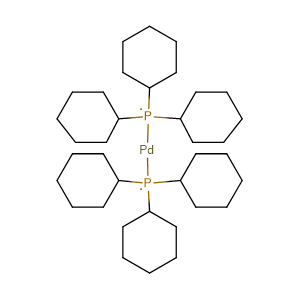
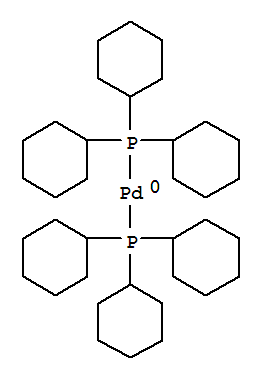
Bis(tricyclohexylphosphine)palladium(0), 98%, Thermo Scientific Chemicals, Quantity: 100 mg | Fisher Scientific
![PDF] Synthesis of pinacol arylboronates via cross-coupling reaction of bis(pinacolato)diboron with chloroarenes catalyzed by palladium(0)–tricyclohexylphosphine complexes | Semantic Scholar PDF] Synthesis of pinacol arylboronates via cross-coupling reaction of bis(pinacolato)diboron with chloroarenes catalyzed by palladium(0)–tricyclohexylphosphine complexes | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4be57a2656bab63a4be8f70bffa3b4b0011d1d07/15-Table1-1.png)
PDF] Synthesis of pinacol arylboronates via cross-coupling reaction of bis(pinacolato)diboron with chloroarenes catalyzed by palladium(0)–tricyclohexylphosphine complexes | Semantic Scholar

Bis(tricyclohexylphosphine)palladium(0), 98%, Thermo Scientific Chemicals, Quantity: 100 mg | Fisher Scientific
![PDF] Synthesis of pinacol arylboronates via cross-coupling reaction of bis(pinacolato)diboron with chloroarenes catalyzed by palladium(0)–tricyclohexylphosphine complexes | Semantic Scholar PDF] Synthesis of pinacol arylboronates via cross-coupling reaction of bis(pinacolato)diboron with chloroarenes catalyzed by palladium(0)–tricyclohexylphosphine complexes | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4be57a2656bab63a4be8f70bffa3b4b0011d1d07/16-Table2-1.png)